BY- Ezhuthachan Mithu Mohan (MSIWM043)
Enzyme Inhibitor: Enzyme inhibitor decreases the rate of reaction by binding to the substrate or decreasing the turnover number. It can be organic or inorganic.
The process which decreases the rate of reaction, by either binding to enzymes or making configurationally changes is known as enzyme inhibition.
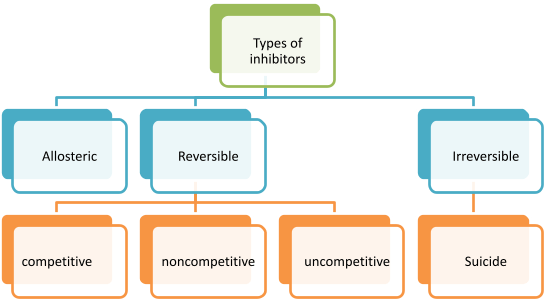
Allosteric Inhibition:
When enzyme poses allosteric side other than active site, allosteric inhibitors bind to the allosteric site causing the configuration change in enzyme, making it less feasible for enzyme to bind with substrate. This type of inhibition is partially reversible, when excess of substrate is added. Km increases and Vmax reduces.
Image source: socratic.org
Allosteric inhibition is mainly of two types
- Positive allosteric inhibition: Which increases the Enzyme activity
- Negative allosteric inhibition: Which decrease the Enzyme activity
Examples of allosteric inhibition
| Phosphofructokinase | Activator: AMP | Inhibitor: ATP and citrate |
| Glycogen phosphorylase | Activator: AMP | Inhibitor: ATP |
End Point inhibition: This type of inhibition is also known as Negative feedback inhibition .It is a specialized form of allosteric inhibition, which controls metabolic pathway by regulating various cellular functions. In this type of inhibition end product when formed in excess, it regulate the pathway by binding to the enzymes in reaction, thus controlling production rate.
Reversible Inhibition:
Weak interaction of inhibitor to enzyme causes reversible inhibition, which can be reverted back to normal by adding excess substrate to compete with inhibitor or by removing inhibitor. This type of inhibition follows Michaelis- Menten rate equation. It has rectangular hyperbolic curve.
| Competitive inhibitor | Noncompetitive inhibitor | Uncompetitive inhibitor |
| When inhibitor binds to the site where substrate binds it causes competitive inhibitor | When inhibitor binds with other site except active site | When inhibitor binds with Enzyme substrate complex, It causes structural distortions |
| These inhibitors are mainly analogues to substrate | These inhibitors are not analogues to substrate | These inhibitors are not analogues to substrate |
| Vmax not changed Km increased and Velocity decreases | Vmax decreases and Km remains unchanged | Vmax and Km decreases |
| Example : Malonate binds Succinate dehydrogenase and competes with Succinate | Example : Ethanol binds with Acid phosphatase | Example: inhibition of placental alkaline phosphatase by phenylalanine |
Irreversible inhibition:
Strong interaction of inhibitor to enzyme causes irreversible inhibition (mostly Covalent interaction). The conformation change caused in Enzyme due to inhibitor is irreversible. Enzyme activity is not regained even by adding or increasing substrate concentration. Vmax decreases and Km is not changed. Suicide inhibition is a specialized form of inhibition, which is also known as mechanism based inactivation. When inhibitor binds to enzyme, it inactivates or complete degradation of enzyme occurs. This type of inhibition does not follows Michaelis- Menten rate equation. It has sigmoidal curve.
Examples: Idoacetate , oxidizing agents etc.
Significance of Enzyme inhibition:
- To study Drug action
- To study efficiency of enzyme
- The interaction of enzyme with substrate can be clearly understood
- Elucidating cellular reactions by accumulation of intermediates
- Identification of catalytic site
- Various drugs used are inhibitors of reaction, so the efficiency of drug and its catalytic function can be clearly known.
.