BY: RAHUL ANDHARIA (MSIWM001)
Introduction:
- DNA in a cell is target for various endogenous and exogenous agents that can damage the base or sugar phosphate backbone.
- It is estimated that each day 104 or 106 lesions are produced in the DNA of a human cell. Hence, this lesions needs to be repaired in order to avoid mutations in the DNA.
- Prokaryotic and Eukaryotic cells have specialized mechanisms to identify and correct various kinds of damage. The rate of this repair depends on the factors such as cell type, age and extracellular environment.
- A cell that is no longer able to repair its DNA damage can enter one of these possible states: senescence (old age), Apoptosis (cell death), and Neoplasia (unregulated cell division).
Types of Damages:
- There are agents like Endogenous and exogenous agents which can damage the DNA.
- There are two major sources of endogenous DNA damage:
- Reaction of components with DNA with extremely reactive metabolites, such as reactive oxygen species (ROS) and reactive nitrogen species (RNS) produced from biochemical pathway.
- Errors in DNA replication or repair by DNA polymerase.
- The main types of damage due to endogenous cellular processes are; single stranded and double stranded breaks, hydrolysis of glycosidic bonds leading to depurination or depyremidation.
- Oxidation of bases due to ROS resulting in products such as 8-oxo-G also occurs due to endogenous cellular processes.
Exogenous agents that can cause damage includes:
- Cosmic ionizing radiations.
- UV solar radiations.
- Fungal and plant toxins.
- Man made mutagenic chemicals like intercalating agents.
- Types of damages generally caused by exogenous agents include; single and double stranded breaks, pyrimidine dimmers, modified bases like ethenobase, oxidized bases.
- Inter and intra strand crosslink’s are also caused by exogenous agents.
DNA repair systems and their mechanisms:
- Direct Reversal of Damage:
- Cells can eliminate three types of base damage by chemically reversing it. This type of repair does not require a template on the complementary strand (normal, non-damaged base).
- Photo reactivation by Photolyase:
- The absorption of UV light by DNA results in the formation of pyrimidine dimmers, mostly thymine dimmers.
- An enzyme called photolyase is activated by energy absorbed from UV light and directly reverses this damage to restore the pyrimidines to their original un-dimerized form.
- Photolyase has two types of chromospheres; FADH in all organisms, Folate- in E.coli and yeast. Photolyase is absent in placental mammals.
Mechanism:
- The photo antenna in photolyase, N5, N10-Ethenyl-tetrahydro-folyl-polyglutamate absorbs a blue light photon at 300-500nm of wavelength.
- It transfers the excitation energy to FADH in the active site of the enzyme.
- The excited flavin (FADH) donates an electron to the pyrimidine dimmer. This creates an unstable dimmer radical.
- The unstable radical undergoes electronic rearrangement to revert to monomeric pyrimidines.
- The electrons are then transferred back to FAD+ to generate FADH.
- Methyl Guanine Methyl Transferase:
- O6 methyl guanine is a common lesion produced by alkylating agents. It is highly mutagenic because methyl-G tends to pair with with T rather than C during replication. These mispairing results in G: C to A: T transition.
- Although alkylating mutagens preferentially modify the guanine base at N7 position, O6 is a major carcinogenic lesion in DNA.
- The DNA adduct formed, is removed by the repair protein O6 methyl guanine methyl transferase through SN2 mechanism. Since it removes the alkyl group from the lesion, this protein is not a true enzyme.
- The methyl acceptor in the protein is a Cysteine.

- Base Excision Repair: (BER)
- In this mechanism, damage is caused to a single nucleotide by oxidation, alkylation, hydrolysis or deamination.
- There are several specific enzymes in BER mechanism, each able to identify a specific type of base lesion.
- These are the DNA N-glycosylases, which can cleave the glycosidic bond between a damaged base and the deoxyribose moiety.
- When glycosylase comes across a damaged base, it hydrolyses the glycosidic bond to generate AP (apurinic/apyrimidinic) sites.
- Now, the AP endonucleases cleaves the phosphodiester bond at 5’ of AP site, leaving 3’ OH group and a 5’ deoxyribose phosphate residue, marking the abasic site.
- This creates a site for binding of DNA polymerase I, which then uses the 3’OH terminus to replace a few nucleotides using the complementary strand as template.
- If only one nucleotide is repaired, it is called as short patch repair. If several nucleotides (2-10) are replaced it is called long patch repair.
- Most of the BER is via short patch repair.
- As the DNA polymerase adds new nucleotides to the 3’end created by AP endonuclease, it displaces the nucleotides 3’ of the nick, leading to Flap.
- This flap is cleaved by deoxyribose phosphatase in short patch repair and by flap endonuclease in long patch repair.
- Finally, the DNA Ligase seals the nick between the patch filled in by DNA polymerase I and the residue exposed by removal of overhanging.

- Nucleotide Excision Repair:(NER)
- Nucleotide excision repair- repairs damage affecting longer stretch of DNA, comprising 2-30 bases. The enzymes in NER can recognize bulky, helix-distorting lesions, intra-strand cross links, oxidative damage as well as single strand breaks.
- NER has two sub pathways; Global genomic NER and Transcription coupled NER.
- NER is critical to survival of all free living organisms; especially those that lack the photolyase system for direct repair of UV induced pyrimidine dimmers.
- In E.Coli the key enzyme complex for NER is the ABC exonucease. This complex comprises three subunits, UVrA, UVrB, and UVrC. The complex is called exonuclease as it catalyses two specific endonucleolytic cleavages, one on either side of lesion.
- This lesion is detected by a complex of UVrA and UVrB proteins which scans the DNA and binds to the site of the lesion.
- Once the lesion is recognized, the UVrA dissociates, leaving behind a tight UVrB-DNA complex.
- Now, UVrC binds to UVrB and mediates two incisions.
- UVrC mediates the incision at 8th phosphodiester bond on the 5’ side.
- UVrB cuts at the 5th phosphodiester bond on the 3’end of the lesion.
- Thus, a 12-13 nucleotide long fragment encompassing the lesion is cut out of the DNA.
- In the next step, the UVrD helicase removes the 12-13 nucleotide fragment by unwinding it away from the dsDNA.
- DNA Ligase seals the nick and complete the process.
- Defects in genes for NER proteins results in genetic disorders like, Xeroderma Pigmentosum, Cokaynes syndrome.

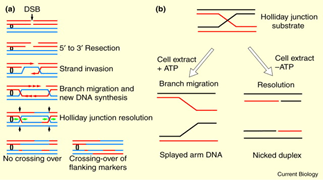
- Homologous Recombination Repair:
- Double strand breaks of DNA double helix can be repaired by homologous recombination repair.
- A DNA cleaving enzyme degrades the broken DNA molecule to generate 3’ end.
- The single strand tails thus, generated will invade the unbroken homologous DNA duplex.
- Invading strand base pairs with its complementary strand present in the other DNA molecule.
- The invading strands with 3’oH group will serve as primers for new DNA synthesis.
- Eventually, the second strand also invades and repairs the DNA from 3’end.
- This will generate two junctions known as Holiday Junctions. These holiday junctions are produced because of Branch migration.
- This recombination intermediate will be further resolved by cleavage.
- Mismatch Repair:
- This repair mechanism recognizes and repairs errors in insertions, deletions, and in mis-incorporation of bases. It plays a vital role in Homeostasis and genomic stability.
- The mismatch is recognized and the DNA is kinked towards major groove. MUTs detects the mismatch with the help of conserved motif, Phe-X-Glu.
- MUTs and MUTL interaction occurs and forms a bridge for other protein complexes.
- Ternary complex is formed by MUTs, MUTL and mismatched DNA.
- MUTS alpha activates EXO1 and removes mismatch base.

DNA repair mechanisms are essential to ensure species survival by enabling faithful inheritance of parental DNA. DNA repair mechanisms if failed to work can lead to cancer and mutations, which may lead to genetic disorders.
