BY: SAI MANOGNA (MSIWM014)
Many leukocyte forms shift from one part of the body to the next. It refers primarily to lymphocytes that circulate in the blood and lymph continuously and travel through other types of leukocytes into the tissues at sites of infection or tissue damage. Not only does this recirculation increase the probability that lymphocytes specific to a specific antigen will encounter that antigen, but it is also crucial for inflammatory response development. The complex response to local injury or other damage is inflammation; it is characterised by redness, heat, swelling, and pain. Various immune system cells and various mediators are involved in inflammation. Without the regulated movement of leukocyte populations, assembling and controlling inflammatory responses would be difficult.
Recirculation of Lymphocytes:
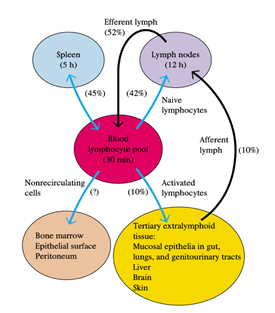
1. Lymphocytes are capable of a remarkable recirculation stage, continually flowing to the different lymphoid organs via the blood and lymph.
2. Almost 45% of all lymphocytes are transferred directly from blood to spleen after a short transit period of approximately 30 minutes in bloodstream, where they live for approximately five hours.
3. Nearly identical numbers of lymphocytes (42%) exit from the blood into separate peripheral lymph nodes, where they live for about 12 hours.
4. A smaller number of lymphocytes of about 10% move into tertiary extra lymphoid tissues by crossing between endothelial cells that line the capillaries.
5. Typically, these tissues have few, lymphoid cells, but during an inflammatory response, they will import them.
6. Skin, pulmonary and genitourinary tract, and various mucosal epithelia of the gastrointestinal, interface with the external environment are known as immunologically active tertiary extra lymphoid tissues.
Mechanism:
1. The mechanism of continuous lymphocyte recirculation causes the antigen to be identified in the maximum number of antigenically committed lymphocytes.
2. An individual lymphocyte can make a complete circuit as much as 1-2 times per day from blood to tissues and back to lymph.
3. Since a particular antigen is recognised by only about one in 105 lymphocytes, a large number of T or B cells will tend to have affected the antigen in a given antigen-presenting cell within a short period in order to produce a specific immune response.
4. By thorough recirculation of lymphocytes, the small percentage of lymphocytes committed to a given antigen makes contact with that antigen when its presence is increased.
5. Factors that control, coordinate, and direct the circulation of lymphocytes and antigen-presenting cells also increase the probability of such contacts profoundly.
Cell Adhesion Molecules (CAMs):
The vascular endothelium controls the passage of blood-borne molecules and leukocytes into the tissues as an essential “gate-keeper.” The cells have to bind to and migrate between the endothelial lining cells of the blood vessels, in order for circulating leukocytes to penetrate inflamed tissue or peripheral lymphoid organs. This mechanism is called extravasation.
1. Endothelial cells release CAMs unique to the leukocyte.
2. Some of these membrane proteins are constitutively expressed; others are only expressed as a response to local cytokine concentrations generated during an inflammatory response.
3. Lymphocytes, monocytes, and granulocytes that recirculate carry receptors that bind to vascular endothelium CAMs, allowing these cells to extravasate into the tissues.
4. CAMs on leukocytes often help to increase the strength of functional interactions between immune system cells. They also play their role in adhesion of leukocyte to vascular endothelial cells.
5. It has been shown that different adhesion molecules contribute to the interaction between Th and B cells, Th and APCs, and CTLs and target cells.
6. A variety of CAMs of endothelial and leukocyte have been cloned and characterised, giving new information about the mechanism of extravasation.
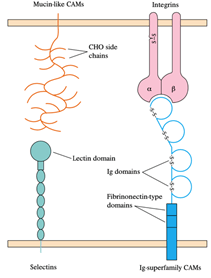
7. Most of these CAMs belong to four protein families: the family of selectin, the family of mucin, the family of integrin, and the superfamily of immunoglobulin (Ig).
| Mucin-like CAMs | Selectins | Ig superfamily CAMs | Integrins |
| GlyCam-1 CD34 PSGL-1 MAdCAM-1 | L-selectin P-selectin E-selectin | ICAM-1,2,3 VCAM-1 LFA-2 (CD2) LFA-3 (CD58) MAdCAM-1 | α4β1 (VLA-4, LPAM-2) α4β7 (LPAM-1) α6β1 (VLA-6) α Lβ 2 (LFA-1) α Mβ 2 (Mac-1) α Xβ 2 (CR4, p150/95) |
SELECTINS:
1. The membrane glycoprotein selectin family has a distal lectin-like domain that allows these molecules to attach to particular groups of carbohydrates.
2. Selectins mainly interact with sialylated carbohydrate moieties, also associated with mucin-like molecules.
3. Three molecules named L, E, and P are part of the selectin family.
4. Most circulating leukocytes express L-selectin, while vascular endothelial cells express E-selectin and P-selectin.
5. The Selectin molecules are responsible for the initial vascular endothelium stickiness of leukocytes.
MUCINS:
1. Mucins are a group of strongly glycosylated serine- and threonine-rich proteins. 2. Their extended structure enables selectins to be presented by sialylated carbohydrate ligands.
3. For instance, sialylated carbohydrates on two mucin-like molecules (CD34 and GlyCAM-1) expressed on specific endothelial cells of lymph nodes are recognised by L-selectin on leukocytes.
4. The mucin-like molecule which is PSGL-1 found on neutrophils interacts with the inflamed endothelium expressed by E- and P-selectin.
INTEGRINS:
1. The integrins are heterodimeric proteins that are expressed by leukocytes consisting of ⍺ and β chain which promote both vascular endothelium adherence and other cell-to-cell interactions.
2. Integrins are classified according to which they comprise a subunit.
3. Different integrins are expressed by different leukocyte populations, allowing these cells to bind to various CAMs along the vascular endothelium which belong to the immunoglobulin superfamily.
3. Some integrins are activated before they bind to their ligands with high affinity.
4. Leukocyte-adhesion deficiency (LAD), and an autosomal recessive disorder shows the significance of integrin molecules in leukocyte extravasation.
5. It is characterised by repeated infections with bacteria and delayed wound healing.
ICAMS:
1. Several adhesion molecules contain a variable number of domains identical to immunoglobulins and are thus categorised within the superfamily of immunoglobulins.
2. ICAM-1, ICAM-2, ICAM-3 and VCAM, which are expressed in vascular endothelial cells and bind to different integrin molecules.
3. Both Ig-like domains and mucin-like domains have an essential cell-adhesion molecule called MAdCAM-1.
4. This molecule is expressed in the endothelium of the mucosa and directs the entry of lymphocytes into the mucosa.
5. It binds to integrins through its domain-like immunoglobulin and selectins through its domain-like mucin.
Extravasation of Neutrophils:
Different cytokines and other inflammatory mediators act upon the local blood vessels by which an inflammatory response develops, inducing increased expression of endothelial CAMs. It is then said to activate or inflame the vascular endothelium. The first cell type of binding to inflamed endothelium and extravasate into the tissues is usually neutrophils. Neutrophils must identify the inflamed endothelium and bind tightly enough so that the flowing blood does not sweep them away. The endothelial layer must then be penetrated by the attached neutrophils and migrate into the underlying tissue. By a similar mechanism, monocytes and eosinophils extravasate, but the best steps for the neutrophil have been identified, so here we concentrate on neutrophils.
It is possible to divide the neutrophil extravasation process into four sequential steps:
(1) rolling processes,
(2) activation by the stimulation of the chemoattractant,
(3) arrest and accession,
(4) migration of transendothelial.

Mechanism:
1. In the first step, via a low-affinity selectin-carbohydrate interaction, neutrophils bind loosely to the endothelium.
2, Cytokines and other mediators act upon the local endothelium during an inflammatory response, inducing expression of the selectin family’s adhesion molecules.
3. These E- and P-selectin molecules bind on the neutrophil membrane or with a sialylated lactose aminoglycan called sialyl Lewis to mucin-like cell-adhesion molecules.
4. This interaction briefly tethers the neutrophil to the endothelial cell, but soon the neutrophil is detached by the sheer force of the circulating blood.
5. Selectin molecules tether the neutrophil on another endothelial cell; this process is replicated so that the neutrophil tumbles end-over-end around the endothelium. This form of binding called rolling
6. different chemoattractants activate it as the neutrophil rolls; these are either permanent features of the surface of the endothelial cell or secreted locally by cells involved in the inflammatory response.
7. Members of a broad family of chemoattractive cytokines called chemokines are amongst the chemoattractants. Interleukin 8 (IL-8) and the macrophage inflammatory protein (MIP-1) are two chemokines involved in the activation process. Not all chemoattractants belong to the category of chemokines.
8. Other chemoattractants include platelet-activating factor (PAF), split-complement products such as C5a, C3a, and C5b67, and various N-formyl peptides produced during infection by breaking down bacterial proteins.
9. Binding to receptors on the neutrophil membrane of these chemoattractants triggers an activating signal mediated by receptor-associated G proteins.
10. This signal causes the integrin molecules in the neutrophil membrane to change conformationally, increasing their affinity with the endothelium Ig-superfamily adhesion molecules.
11. Subsequent contact between integrins and CAMs of the Ig superfamily stabilises the neutrophil’s adhesion to the endothelial cell, making it possible for the cell to bind tightly to the endothelial cell.
12. The neutrophil subsequently migrates into the tissues via the vessel wall.
The further steps in transendothelial migration and how it is guided are still largely unknown; they can be mediated by further activation or by a separate migration stimulus by chemoattractants and subsequent integrin-Ig-superfamily interactions.

