BY: Ria Fazulbhoy (MSIWM031)
Introduction
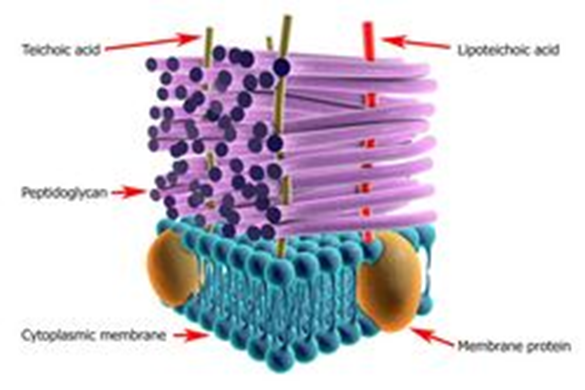
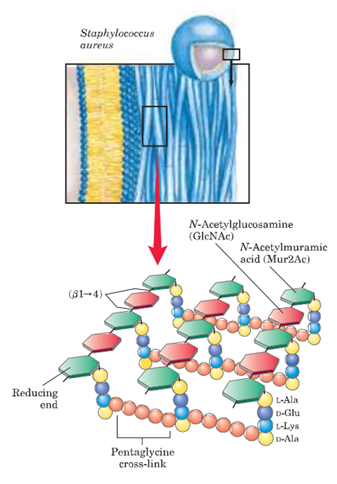
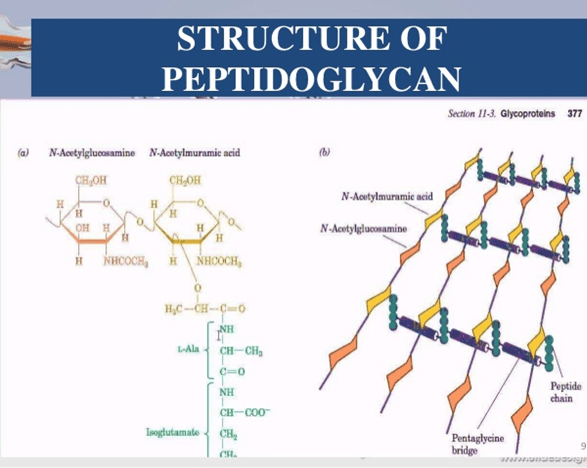
Bacteria require a thick, rigid extracellular wall that protects them from osmotic lysis. There is a structure known as the peptidoglycan (also known as muerin) which protects the bacteria and gives the bacterial envelopes their strength and rigidity. Peptidoglycan is a polymer made up of amino acids and sugars which forms a mesh like layer. It is a linear, alternating copolymer of N Acetylglucosamine (GlcNAc) and N-acetylmuramic acid (Mur2Ac), linked by beta 1->4 glycosidic bonds, which is crosslinked by short peptides attached to the Mur2Ac. Both GlcNAc
and Mur2Ac are activated by attachment of a uridine at their anomeric carbons during the assembly of the polymer of this complex macromolecule.
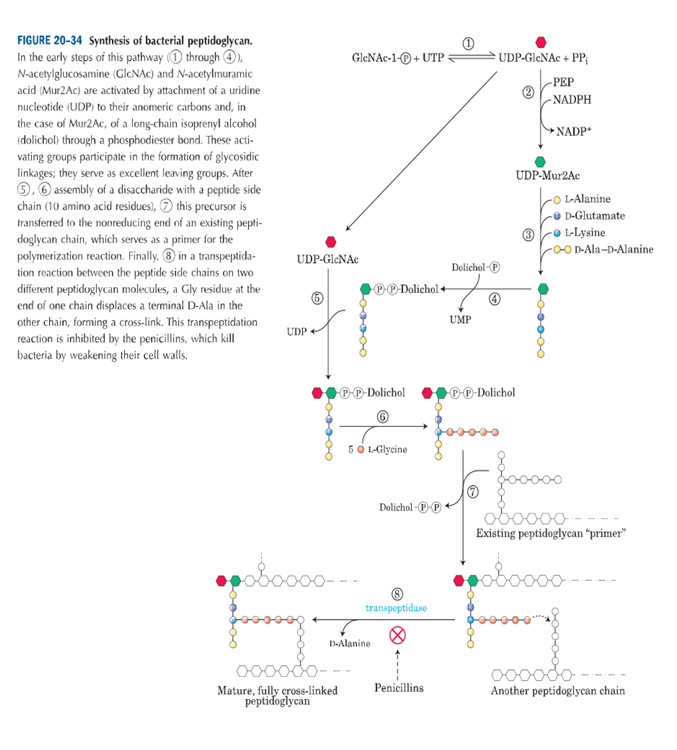
Bacterial peptidoglycan synthesis
The peptidoglycan outside the plasma membrane of bacteria is synthesised in the following manner:
- In the first step, GlcNAc 1-phosphate condenses with UTP to form UDP-GlcNAc.
- UDP-GlcNAc then reacts with phosphoenolpyruvate to form UDP-Mur2Ac, with NADPH present.
- To this, 5 amino acids are added to form Mur2Ac pentapeptide moiety.
- The Mur2Ac pentapeptide moiety is made from amino acids L alanine, D glutamate, L lysine, D ala D alanine.
- This pentapeptide moiety is then transferred to membrane lipoid dolichol, a long chain isoprenoid alcohol, from the uridine nucleotide.
- UDP GlcNAc also donates a GlcNAc.
- In a number of bacteria, five glycines are then added in peptide linkage to the amino group of the Lys residue of the pentapeptide.
- After this, the disaccharide decapeptide which is formed, is added to the non reducing end of an already existing peptidoglycan molecule. Dolichol leaves the macromolecule in this step.
- A transpeptidation reaction occurs in which there is crosslinking of the adjacent polysaccharide chain. This step is catalysed with the help of the enzyme transpeptidase.
- Thus, a huge, strong macromolecule is formed which contributes to the macromolecular wall surrounding the bacterial cell.
- Most of the effective antibiotics that are used today against bacteria, have a mode of action by inhibiting the reactions which are involved in the synthesis of bacterial peptidoglycan.
- Numerous other oligosaccharides and polysaccharides are synthesized by similar routes, where sugars are activated for subsequent reactions by attachment to nucleotides
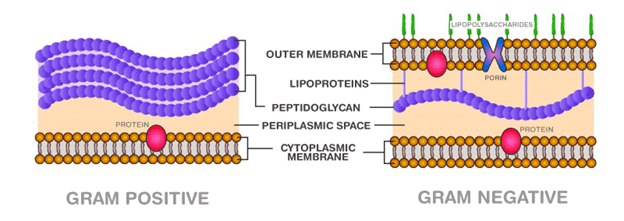
Difference of peptidoglycan in gram positive and gram negative bacteria
Importance of peptidoglycan in bacteria
The peptidoglycan which surrounds the bacteria is very important, and sometimes is essential for their survival. They have 2 main functions:
- To counteract and maintain the osmotic pressure of the cell. If the peptidoglycan is absent, the bacteria undergoes very abrupt osmotic lysis. Hence, a lot of antibiotics target the peptidoglycan of the bacteria, since it plays a crucial part in the maintenance of osmotic pressure and protection of the bacterial cell. These antibiotics inhibit the synthesis of peptidoglycan in bacteria and initiate osmotic lysis of it.
- Another very important function of the peptidoglycan is the regulation of the molecules entering and leaving the bacterial cell. The peptidoglycan regulates the diffusion of cells which play important roles like division of the cell and anchoring the cell wall (eg : teichoic acid)
- Some bacteria also release peptidoglycan fragments, which play an important role in cell to cell communication.


There’s definately a great deal to know about this topic. I really like all the points you made.
LikeLike
I am glad to be a visitant of this staring web blog! , appreciate it for this rare information! .
LikeLike